INTRODUCTION
Urinary Tract Infections (UTIs) are the most prevalent bacterial infectious diseases encountered in both community and hospital settings, imposing huge impact on global health1. UTIs are a widespread health concern in developing countries, and Bhutan is no exception. According to Bhutan’s Annual Health Report of 2020, there were 35,657 cases related to kidney and urinary tract diseases, of which 1,901 were specifically identified as cystitis2. These statistics emphasize the need for a comprehensive understanding and effective management of UTIs.
UTIs present with symptoms like dysuria, increased frequency, urgency, altered urine color, flank pain and, in some cases, fever3. Apart from clinical symptoms, UTIs are diagnosed by urinalysis, either with a urine dipstick or microscopy, and urine culture. However, the gold standard for confirming uropathogens is urine culture with antibiotic sensitivity testing4,5.
The emergence of antibiotic resistance is a global concern, varying across regions based on distinct antibiotic regimens. Inaccurate diagnoses and the use of antibiotics based solely on symptoms contribute to the rising rates of antibiotic resistance in microbes causing UTIs4. This emergence of antibiotic resistance not only complicates disease management but also poses a significant public health concern including increased healthcare expenditures. Complications such as renal scarring, often observed after UTIs in infancy, can lead to hypertension, end stage renal disease, and proteinuria, further exacerbating the health burden6.
As UTIs are a significant health challenge, this study aimed to provide updates on the local antibiotic sensitivity patterns of uropathogens, which could guide healthcare workers towards the effective therapeutic management of UTIs in low resource settings where culture facilities are not available.
METHODS
Study Design and Setting
This cross-sectional descriptive study was conducted at the General Outpatient Department (GOPD) of Jigme Dorji Wangchuck National Referral Hospital in Thimphu, from September 2021 to August 2022. The GOPD has five clinicians who offer clinical services to patients of all ages and specialties. Ethical clearance was obtained from the Institutional Review Board (IRB) [Ref. No. IRB/Approval/PN21-015/2021/521] dated 21/09/2021.
Study Population and Sample Size
The study included patients aged 13 years and above with UTIs symptoms. Patients with urinary symptoms who were already undergoing antibiotic treatment were excluded. Records maintained at the GOPD revealed that 1000 new cases with UTI symptoms were treated over a period of six months from January to June 2020. Therefore, we estimated around 2000 patients with UTI symptoms to visit the GOPD during our study period. A sample size of 355 was calculated by using Open Epi, Version 3, taking into consideration a 95% confidence interval.
Sampling technique and data collection
All the clinicians in the GOPD were sensitized on the study’s aims, inclusion and exclusion criteria. Data collection took place on Mondays and Thursdays via convenience sampling until the required sample size was met. On the designated data collection days, clinicians in various chambers of the GOPD directed potential participants to the principal investigator. Participants were enrolled once they provided informed written consent. After enrollment, the principal investigator gathered data using an interviewer administered questionnaire. The questionnaire consisted of three sections. The first section gathered demographic and clinical details including age, sex, marital status, history of previous UTIs, and the presence of any comorbidities. The second section focused on symptoms of UTI, while the third section included the results of urine microscopic analysis, urine culture and antibiotic sensitivity, which were recorded once the results were available. All participants were instructed to collect a clean catch midstream urine sample in two sterile wide-mouth containers, which were submitted to the laboratory for microscopic examination and culture.
Sample processing a) Microscopic examination
Urine samples were processed within 2 hours of receipt at the lab, whenever feasible. Otherwise, they were stored in the refrigerator at 2°C to 8°C for up to 4 hours before processing. For processing, 10 mL of urine was centrifuged for 3 minutes at 1800 revolutions per minute. One drop of the centrifuged mixed sediment was poured onto a clean, labeled slide for examination. Microscopic examination showing White Blood Cells (WBCs) of 0-5 cells/high power field (HPF) were considered normal, while the presence of more than 5 cells/HPF indicated infection.
b) Sample culture processing
Urine culture samples submitted to the microbiology unit were processed within two hours. When not feasible, the samples were stored at 2°C to 8°C in a refrigerator until processed. About 1µL of urine was streaked in a cone-shaped manner using a standard loop on a culture plate containing cystine-lactose-electrolytedeficient (CLED) media. The plates were immediately incubated at 37°C under aerobic conditions overnight, and the presence of growth, extent of growth, and types of colonies were observed. Results were interpreted as follows:
• ‘Negative’ if no growth was seen after 24 hours of incubation.
• ‘Insignificant growth’ if less than 10 Colony-Forming Unit (CFUs)/microliter.
• ‘Probably significant growth’ if 10-100 CFUs/microliter and pure growth.
• ‘Significant growth’ if ≥ 100 CFUs/microliter and pure growth.
• ‘Mixed growth’ if two or more bacteria were seen.
Urine culture plates showing growth were further subjected to the Triple Sugar Iron Agar (TSIA) test for the identification of organism.
c) Antibiotic sensitivity testing (ABST)
Bacterial isolates then underwent ABST using the disk diffusion method according to the Clinical and Laboratory Standards Institute (CLSI) guideline. Antibiotics disks containing commonly used antibiotics in the OPD like amoxicillin 10mcg, ampicillin 10mcg, nitrofurantoin 300mcg, norfloxacin 10mcg, trimethoprim-sulfamethoxazole 25mcg, ceftriaxone 10mcg, and gentamicin 10mcg, were used. Higher generations of antibiotic disks were used when atypical organisms were isolated.
Statistical analysis
Data entry and analysis were performed with Microsoft Access and Statistical Package for Social Science (SPSS) version 25 respectively. Descriptive analysis was used to characterize sociodemographic features, clinical symptoms, uropathogens isolated, and antibiotic sensitivity. The Pearson Chi-square test was used to assess associations between culture positive UTI and associated factors. Binomial logistic regression was used to calculate Odds Ratio (OR) at a 95% confidence interval. Adjusted Odds Ratio (AOR) was calculated for statistically significant variables. A significance level of p < 0.05 was established.
RESULTS
Socio-demographic characteristics
Of the 355 participants clinically diagnosed with UTI, statistical analysis was conducted on 351 participants who provided urine samples for microscopy and culture sensitivity tests. Participants in the age range of 21-45 years comprised the largest proportion (241, 68.7%). The majority of participants were female (273, 77.8%) and married (242, 69%). As depicted in Table 1, most participants did not have a prior documented history of UTI (219, 62.4%) and did not have comorbidities (303, 86.3%).
Common clinical presentations among participants
Dysuria (328, 93.4%), increased frequency of micturition (324, 92.3%), and lower abdominal pain or flank pain (323, 92%) were reported as the most common symptoms. Fever was the least common symptom, reported by only 29.9% (105) of the enrolled participants. The frequency of occurrence of other symptoms are as depicted in Figure 1.
Table 1. Socio-demographic profile of patients clinically diag-nosed as UTI during the study period N=351
|
Characteristics |
n (%) |
|
Age (years) |
|
|
13 – 20 21 – 45 46 – 65 >65 |
25 (7.1) |
|
241 (68.7) |
|
|
58 (16.5) |
|
|
27 (7.7) |
|
|
Gender |
|
|
Male Female |
78 (22.2) |
|
273 (77.8) |
|
|
Marital status |
|
|
Married Unmarried In a relationship Others |
242 (69.0) |
|
57 (16.2) |
|
|
42 (12.0) |
|
|
10 (2.8) |
|
|
Documented previous UTI |
|
|
Yes No |
132 (37.6) |
|
219 (62.4) |
|
|
Documented comorbidities |
|
|
Yes No |
48 (13.7) |
|
303 (86.3) |
Culture positive UTIs
The overall urine culture positivity rate among the 351 patients was 50.53%. The highest culture positivity was observed in females (164, 60.7%) and those in the age group of 46 – 65 years (35, 60.34%). The majority (94, 71.21%) of participants with a documented history of previous UTI had positive urine cultures. Participants with comorbidities exhibited higher culture positivity (28, 58.33%) compared to those without comorbidities.
Associations of culture positive UTIs
Univariable analysis demonstrated that the participants above the age of 21 years, females, married participants, and those with a previous history of UTI had a statistically significant association (p < 0.05) with culture-positive UTI. Further analysis with binomial logistic regression revealed that patients aged between 46 to 65 years had higher odds of having a culture-positive UTI (AOR 1.93, CI 1.30 – 2.86, p ≤ 0.001). As depicted in Table 2, females had an eight times higher odds of culture positive UTI compared to males (AOR 7.99, CI 3.90 – 16.4, p ≤ 0.001), and participants with a previous history of UTI had nearly three times higher odds of culture positive UTI (AOR 2.77, 1.68 – 4.56, CI 95%, p ≤ 0.001).
Table 2. Predictors of culture positive UTI amongst participants presenting to the general OPD during the study period n=351
|
Characteristics |
Positive urine culture |
Negative urine culture |
Crude OR |
P value |
aOR |
P value (95% CI) |
|
Age group (years) |
||||||
|
13 – 20 |
3 |
22 |
Ref |
|
||
|
21 – 45 |
124 |
117 |
7.4 |
0.001 |
|
|
|
46 – 65 |
35 |
23 |
10.45 |
0.001 |
1.93 |
0.001 [1.30 – 2.86] |
|
>66 |
15 |
12 |
8.48 |
0.001 |
|
|
|
Gender |
||||||
|
Male |
13 |
65 |
Ref |
|
||
|
Female |
164 |
109 |
7.42 |
0 |
7.99 |
0.000 [3.90 – 16.40] |
|
Marital status |
||||||
|
Unmarried |
18 |
39 |
Ref |
|
||
|
Married |
135 |
107 |
2.71 |
0.001 |
1.08 |
0.687 [0.73 – 1.61] |
|
In a relationship |
18 |
24 |
1.62 |
0.25 |
|
|
|
Others |
6 |
4 |
3.15 |
0.08 |
|
|
|
Previous history of UTI |
||||||
|
No |
83 |
136 |
Ref |
|
||
|
Yes |
94 |
38 |
4.03 |
0 |
2.77 |
0.000 [1.68 – 4.56] |
|
Co-morbidities |
||||||
|
No |
149 |
154 |
Ref |
|
||
|
Yes |
28 |
20 |
1.44 |
0.238 |
|
|
Comparing urine microscopy and culture in diagnosing UTI
Sensitivity, specificity, positive predictive value (PPV) and negative predictive value (NPV) were calculated to compare urine microscopy with urine culture results. Of the 202 positive urine microscopy samples, 156 (77.2%) turned out to have a positive urine culture. Of the remaining 128 negative urine microscopy samples, 21 (16.4%) had a positive urine culture as depicted in Table 3. Urine microscopy had a sensitivity of 88.1% and specificity of 73.6% in diagnosing UTIs. The PPV and NPV of urine microscopy were 77.2% and 86.0% respectively.
Table 3. Comparison of urine microscopy and urine culture
|
Urine Microscopy |
Urine Culture |
Total |
|
|
Positive |
Negative |
||
|
Positive |
156 |
46 |
202 |
|
Negative |
21 |
128 |
149 |
|
Total |
177 |
174 |
351 |
Types of uropathogens identified in urine culture
The most common isolated pathogen was Escherichia coli (150, 84.7%). As depicted in Figure 2, Klebsiella pneumoniae, Streptococcus saphrophyticus, Streptococcus pneumoniae, and Enterococcus sp., constituted the other uropathogens identified via urine culture.
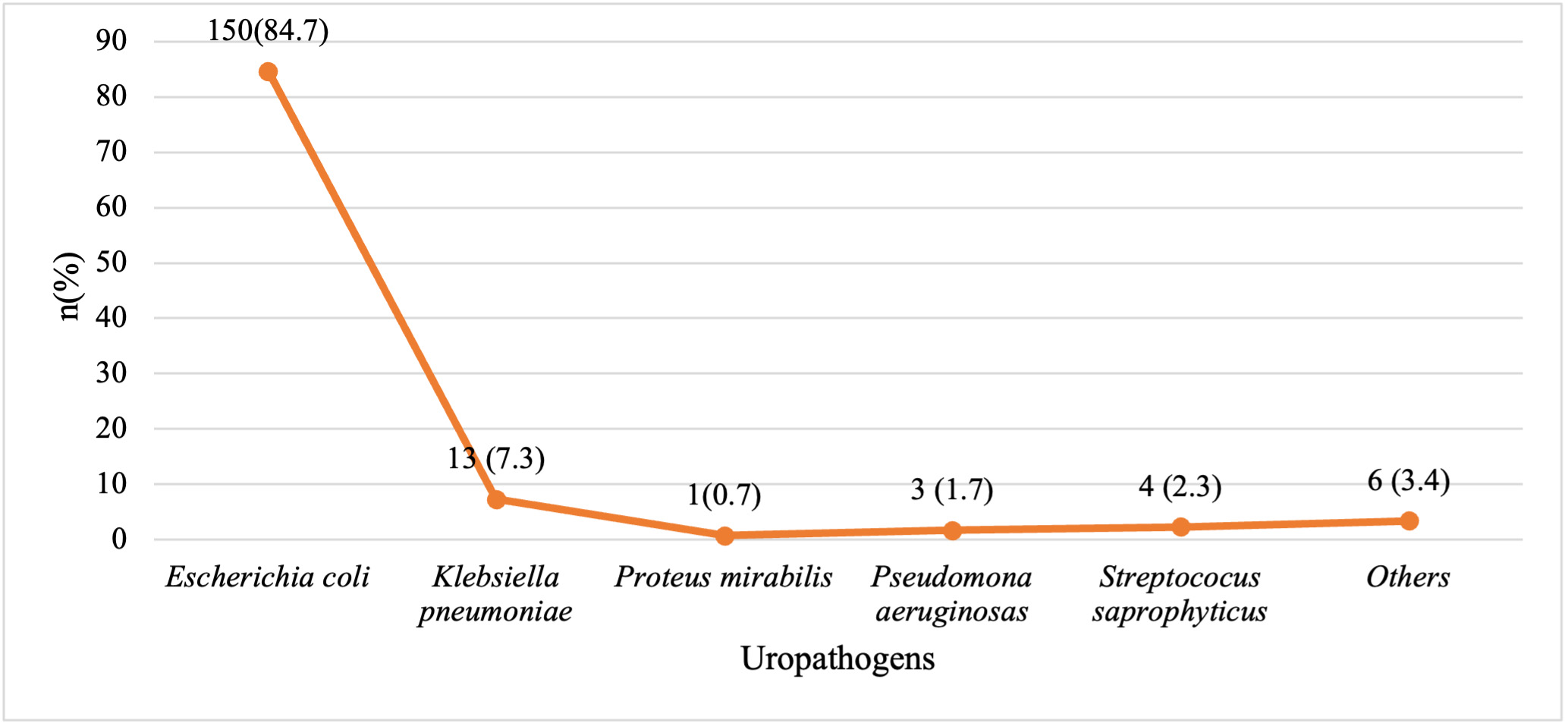
Figure 2. Common Uropathogens isolated in the urine culture of study participants
Antibiotic Sensitivity Pattern
Most of the Escherichia coli isolates were sensitive to nitrofurantoin and gentamicin while the least number of isolates were sensitive to amoxicillin (37.3%) and ampicillin (29.3%). Types of uropathogens identified in urine culture K. pneumoniae was 100% sensitive to gentamicin and 84.6% The most common isolated pathogen was Escherichia coli to nitrofurantoin, with varying sensitivities to other antibiotics. (150, 84.7%). As depicted in Figure 2, Klebsiella pneumoniae, Pseudomonas aeruginosa showed varying sensitivity to different Streptococcus saphrophyticus, Streptococcus pneumoniae, and antibiotics. Table 4 depicts the resistance patterns of the various Enterococcus sp., constituted the other uropathogens identified uropathogens isolated to the commonly used antibiotics in the via urine culture. OPD.
Table 4. Antibiotic sensitivity pattern of uropathogens isolated from participants presenting with UTI symptoms to the general OPD during the study period
|
Uropathogens
|
Antibiotics n(%) |
|||||||
|
AMP |
AMX |
TMT-SMX |
NOR |
NIT |
CTX |
GEN |
Remarks |
|
|
Escherichia coli (n=150) |
44(29.33) |
56(37.33) |
85(56.67) |
110(73.33) |
143(95.33) |
92(61.33) |
143(95.33) |
1 isolate was sensitive to piperacillin, amikacin & meropenem |
|
Klebsiella pneumoniae (n=13) |
X |
X |
10(76.92) |
9(69.23) |
11(84.61) |
10(76.92) |
13(100) |
|
|
Proteus mirabilis (n=1) |
1(100) |
1(100) |
1(100) |
1(100) |
X |
1(100) |
1(100) |
|
|
Pseudomonas aeruginosas (n=3) |
X |
X |
X |
1(33.33) |
1(33.33) |
X |
1(33.33) |
All isolates sensitive to piperacillin, cefazolin, ceftazidime |
|
Streptococcus saprophyticus (n=4) |
X |
X |
3(75) |
4(100) |
4(100) |
X |
X |
All isolates sensitive to penicillin G, cefotaxime & polymyxin |
|
Enterococcus Sp. (n=1) |
X |
X |
X |
X |
1(100) |
X |
X |
Sensitive to penicillin G |
|
Streptococcus pneumoniae(n=1) |
X |
X |
X |
1(100) |
1(100) |
X |
1(100) |
|
|
Others (n=4) |
X |
X |
X |
X |
X |
X |
X |
Mixed growth |
AMP=Ampicillin, AMOX=Amoxicillin, TMT-SMX=trimethoprim-sulphamethoxazole, NOR=Norfloxacin, NIT=Nitrofurantoin, CTX=Ceftriaxone, GEN=Gentamicin, X=Either not sensitive or ABST not tested